WACCABUC, N.Y. - For entrepreneurs like Joe Rosenberg, a once-a-century pandemic presented a unique opportunity to make l...
Problem
Catastrophic global shortage of the N95 respirator: an expensive, uncomfortable and wasteful half-century-old design

Every day, healthcare professionals and frontline workers have to deal with Personal Protective Equipment (PPE) that gets in the way of their comfort, their breathing, and their communication. Disposable N95 respirators currently on the market date back to the 1960's, irritate skin, fog up glasses, force re-breathing of toxic carbon dioxide and create barriers between essential workers and their patients and customers. Not to mention, these devices create over 100,000 tons of landfill waste per year and can cost hospitals over $1,000 per employee annually.
We deserve better.
Solution
Meet Canopy: The supremely comfortable, transparent, reusable, and affordable N95 alternative

Since late March 2020 a team of entrepreneurs, material scientists, biomedical engineers, industrial designers, clinicians, hospital administrators, and former FDA officials has been quietly working to develop and commercialize Canopy, the next-generation reusable respirator. Canopy is led by alumni from top research institutions and innovation hubs including:

(Canopy is not affiliated with, nor endorsed by, the above institutions)
Shipping in Q1 2021, every aspect of Canopy was developed based on direct feedback from healthcare professionals and frontline workers. Over the past seven months our team of Johns-Hopkins-trained biomedical engineers and former Nike industrial designers brought the concept to life.
Finally, Canopy built a supply chain of some of the world's largest, most sophisticated medical device and filtration contract manufacturers and logistics providers to rapidly deploy Canopy on a mass-scale.
Canopy’s patent-pending system solves for the top shortcomings of current N95 respirators. Most importantly, Canopy's novel filter design doesn't use globally-scarce meltblown textiles.
- Cost: Substantially reduced total cost of ownership can save hospitals thousands of dollars per year per employee compared to disposable N95s
- Availability: Canopy does not rely on globally-scarce meltblown filters, and has partnered with the world's largest sterile air filter OEM to manufacture our proprietary filtration solution
Comfort: Silicone face seal does not cause skin abrasions
Breathability: Canopy’s novel, proprietary filter has 3x the surface area of a disposable N95
Communication: Canopy is transparent, enabling lip-reading and smiling
Waste: Converting to Canopy eliminates up to 100 lbs. of annual landfill waste per user
Durability: Canopy is sterilizable, dishwasher safe, and can be sanitized with any hospital-grade disinfectant wipe
Safety: Canopy is a 'half-face' respirator with a seal that goes under the chin and has wings that cover the cheeks, enabling talking/smiling vs. less reliable 'quarter-face' competitors that rest on the chin and run the risk of a broken seal when talking or smiling.
Product
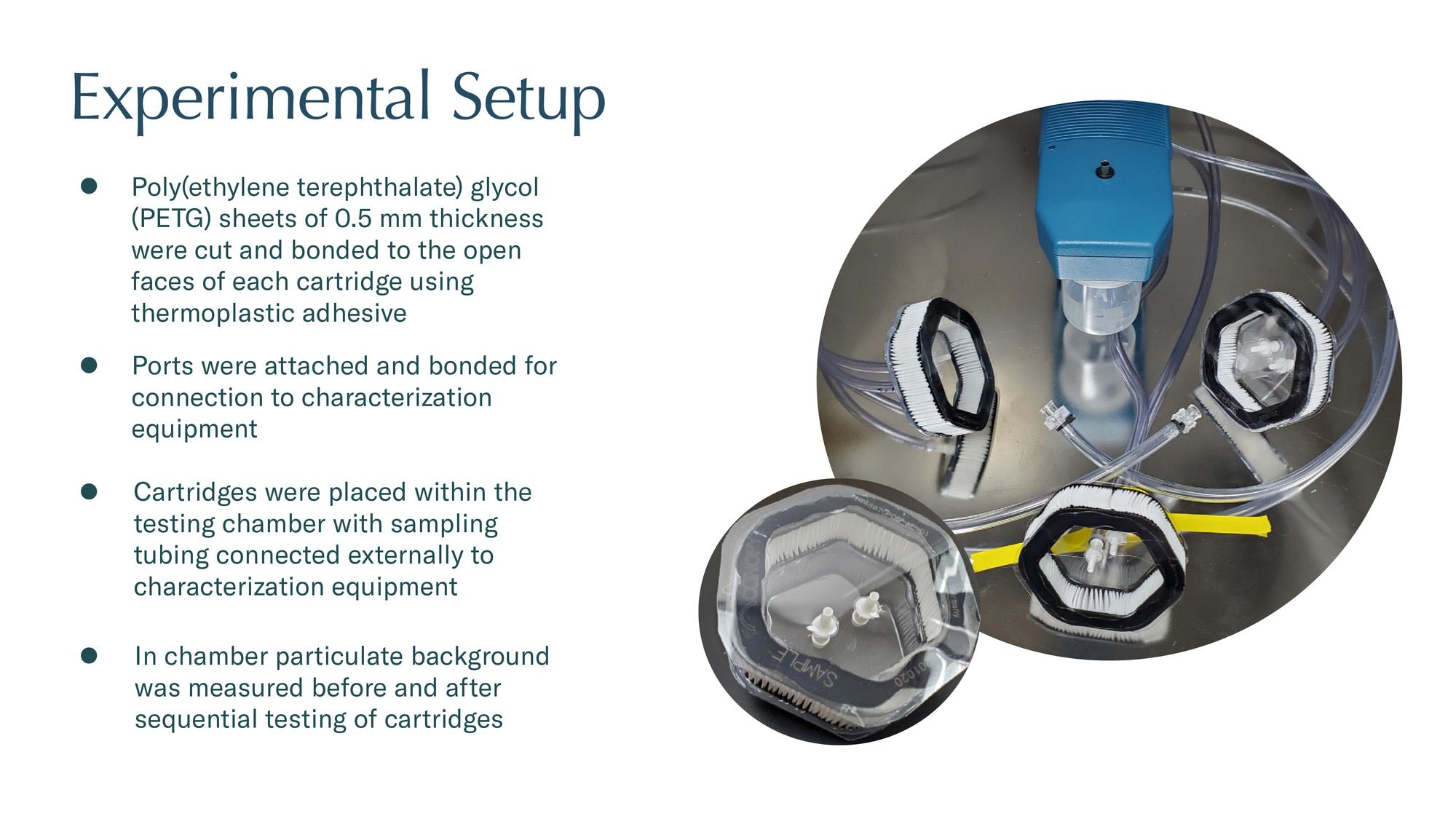
Clinician-designed, hospital-grade respirator with proprietary replacement filter
Shipping in Q1 2021, every aspect of Canopy was developed based on direct feedback from healthcare professionals and frontline workers. Over the past seven months our team of Johns-Hopkins-trained biomedical engineers and former Nike industrial designers brought the concept to life. Finally, Canopy built a supply chain of some of the world's largest, most sophisticated medical device and filtration contract manufacturers and logistics providers to rapidly deploy Canopy on a mass-scale.
Patent-pending filter: Designed by a team with over 100 patents in medical filtration, Canopy’s radially-pleated, patent-pending filter has 3x the surface area of a standard N95, giving users the space to breathe easily. Importantly, Canopy’s filter does not rely on electrostatically-charged meltblown filters. Our raw materials are not in short supply.
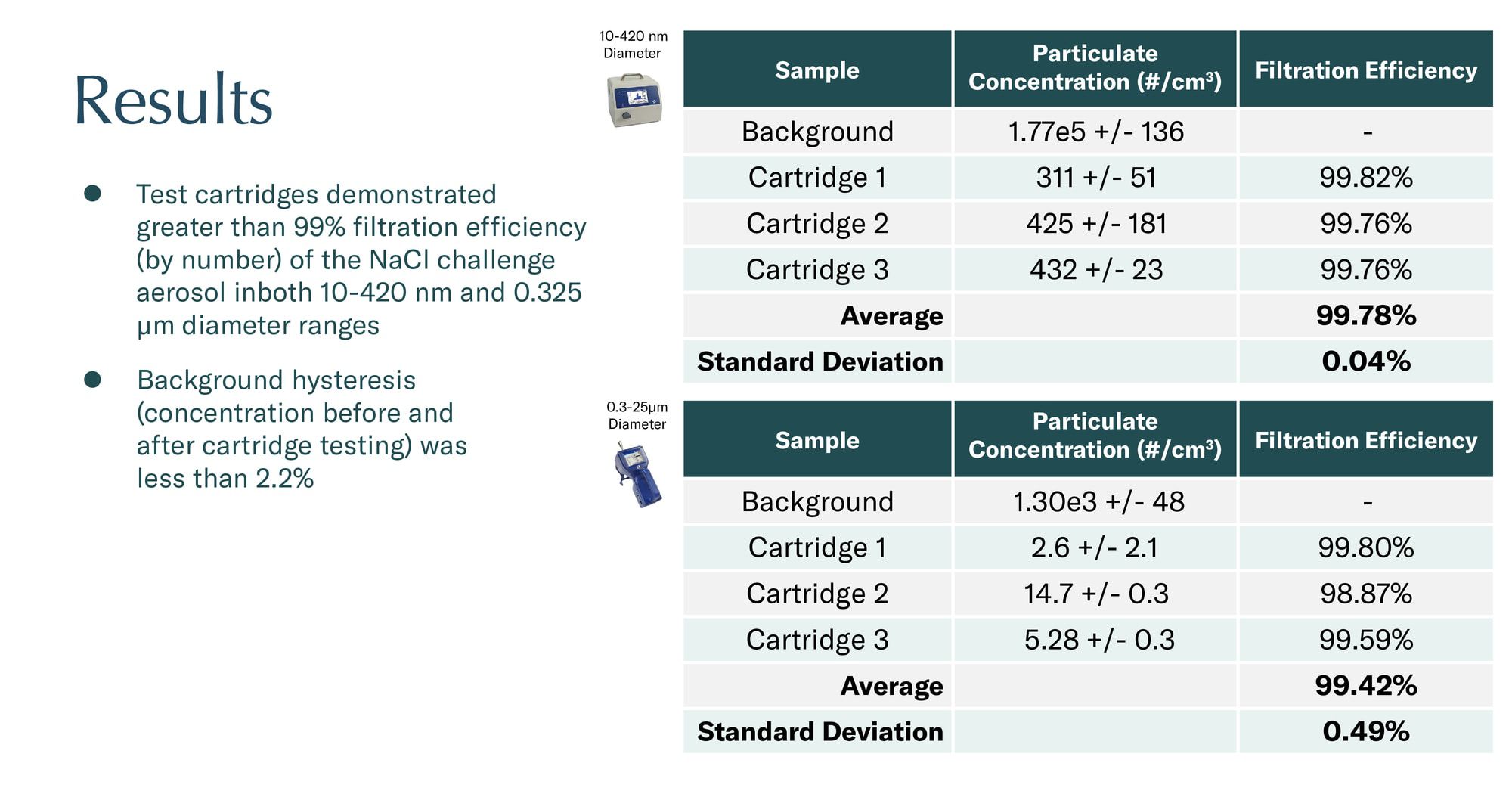
Up to 99.8% filtration efficiency under certain conditions in third-party testing conducted by Drs. Rob MacCuspie, Cary Hill and Matthew Hull at NanoSafe, Inc.
 Transparent front lens: Canopy’s transparent front lens and permanent anti-fog coat means users can interact with patients, customers, friends, and family in a way that allows them to see and be seen, express empathy, smile, and communicate effectively.
Transparent front lens: Canopy’s transparent front lens and permanent anti-fog coat means users can interact with patients, customers, friends, and family in a way that allows them to see and be seen, express empathy, smile, and communicate effectively.
Biocompatible silicone seal: Canopy's form-fitting silicone seal is soft on the skin and snug on the face, guarding users from exposure without leaving “bridge of honor” skin abrasions behind. Each respirator also comes with easy-to-adjust neck straps and a headpiece so comfortable users forget it’s there.
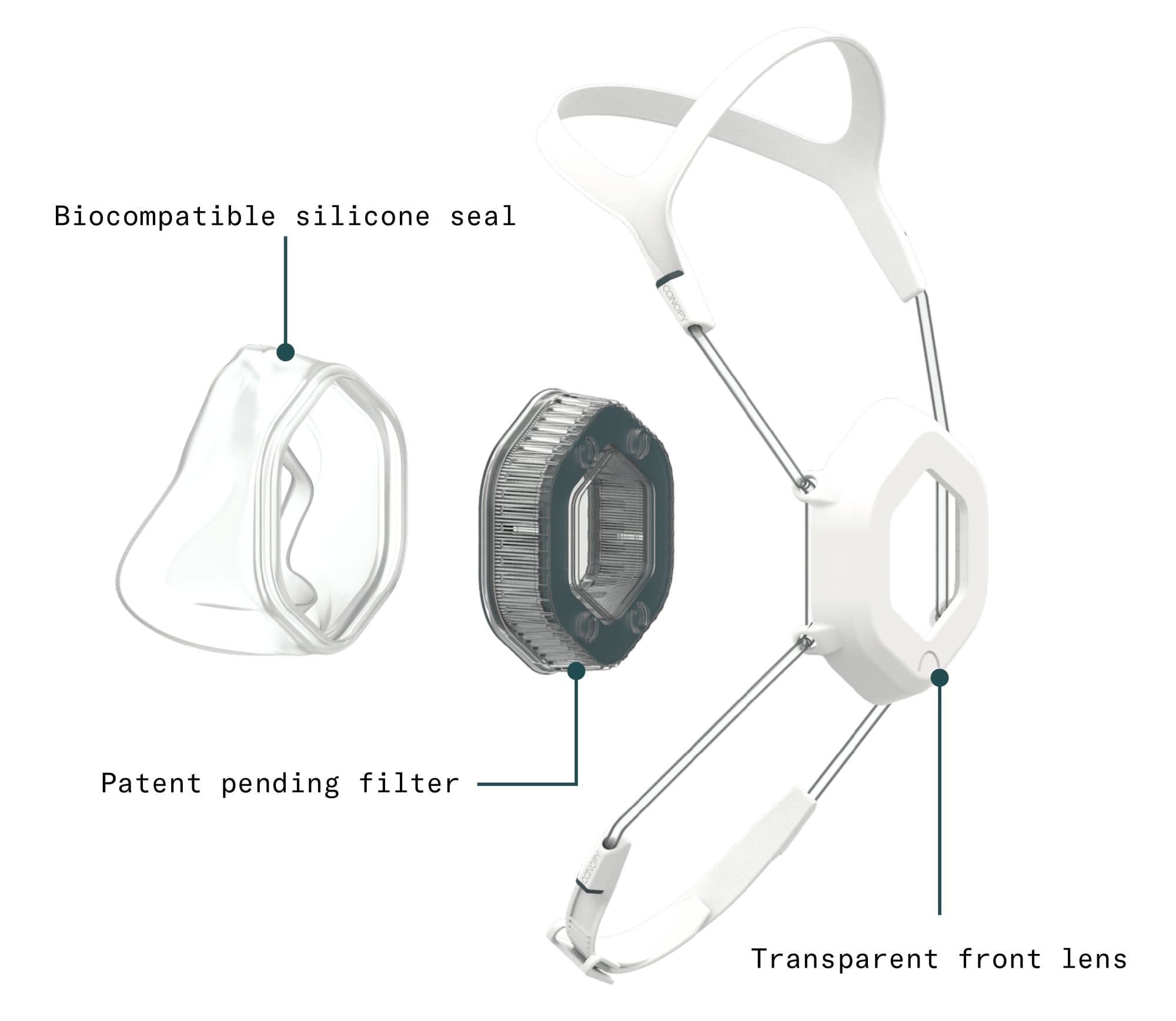
Durable yet comfortable materials: Canopy is made from durable, high-performance polymers that are simple to wipe down with common disinfectants, enabling easy cleaning and sustainable re-use.
Mass-scale US based supply chain: All but two of Canopy’s components are made in the United States and assembled at one of the country's largest ISO 13485 / 9001 medical contract manufacturing facilities.
No metal parts: Canopy can be worn anywhere in the hospital, even near X-Ray and MRI machines.
Utility Patent: Canopy was accepted into the USPTO's COVID "TrackOne" fast track program. We've received our office action / response and will be receiving a full patent in mid 2021.
Customization: Corporate branding and custom colors available by request.

Product Roadmap: Canopy has developed working prototypes of a consumer-grade respirator variant along with a line of interchangeable filters that can be stockpiled for Chemical, Biological, Radiological and Nuclear hazards. We’ve also begun R&D and partnerships work for virus-detecting and virucidal filters.
Short term roadmap:
- Integrated face shield (ETA Q1 2021)

- Fabric strapping system (ETA Q1 2021)

- White-labeled, branded cleaning solutions (wipes, tablets, sprays)
- Full suite of interchangeable N, P, & R rated filters for CBRN hazard stockpiling
Traction
Fulfilling initial shipments and launching hospital pilots in Q1 2021
While operating in stealth mode, Canopy has completed tooling and validation testing, has signed multi-year agreements with some of the world's largest OEM medical device and filtration manufacturers. Our supply chain and global fulfillment infrastructure is ready to begin fulfilling orders in January. Canopy can fulfill $50MM in 2021 order volume without making additional capital investment in production or fulfillment infrastructure.

Northwell Health, New York’s largest health system, will lead an evaluation comparing Canopy against incumbent disposable N95s to demonstrate key benefits such as comfort, breathability, patient experience and economic value. Northwell’s Emergency Medicine Research Consortium developed the protocol in collaboration with the Canopy team.
“Given the significant limitations, drawbacks and shortages of existing PPE, we are eager for our staff to begin testing Canopy in the clinical setting. It cannot come soon enough.” --Dr. Yves Duroseau MD MPH, Chief of Emergency Medicine at Lenox Hill Hospital, part of Northwell Health.
Canopy is also working with researchers at Emory University to evaluate the impact of clear respirators on patient anxiety levels.
Prior to officially launching we’ve received tremendous inbound interest, sample requests and purchase requests from hospital procurement officers, epidemiologists, doctors, nurses, medical leaders, and government officials from institutions including:

Canopy has entered into a strategic partnership with a Access Strategy Partners, Inc. (ASPI), a sales organization led by executive-level veterans from both the buy and sell side of the healthcare procurement market. The ASPI collectively has billions of dollars worth of medical device sales and purchasing experience, and will be leading our enterprise healthcare sales strategy.
In addition to our enterprise sales, Canopy's product suite will be available through our owned and operated e-commerce website, and Canopy "Starter Kits" will be available on across mass-distribution platforms including:

Customers
Taking care of the people who take care of us
Canopy was created in response to a mass shortage of N95 respirators at the onset of COVID-19. Relying on the expertise of healthcare professionals, we designed Canopy so that anyone on the front lines can do their job without the fear of putting themselves or their loved ones at risk.
For those of us who don’t work on the front lines, we too have seen the benefits of having a comfortable and reusable mask to protect ourselves, and the people we love, from fast-spreading illness, wildfire smoke, and poor air quality.

Business model
Hybrid B2B & B2C strategy with recurring subscription
The Canopy team has developed a highly capital-efficient operating model that ensures product and operational excellence through strategic partnerships with best-in-class sales, mass-scale contract manufacturing, third-party logistics and back-office service providers. These partnerships are structured primarily on a variable cost or commission basis, allowing Canopy to remain lean while ensuring supply chain stability and that the resources to support rapid growth are available on demand.
As a company founded in response to the COVID-19 Pandemic, Canopy is not saddled with traditional overhead such as office leases or large travel budgets. Our profitable unit-economics yield strong contribution margins at all sales volumes, and our razor / razor-blade model ensures recurring revenue streams across customer segments.
We expect to become cash flow positive and to begin making initial distributions to investors in 2021, while maintaining a sufficient capital position to meet demand and fuel growth in the form of strategic product roadmap development and geographic expansion.
Subscription Model: Enterprise customers receive preferred pricing with high volume orders and annual supply agreements to ensure constant PPE compliance. Online self-purchasers can replenish filters as desired or sign up for subscribe-and-save auto-renewals.
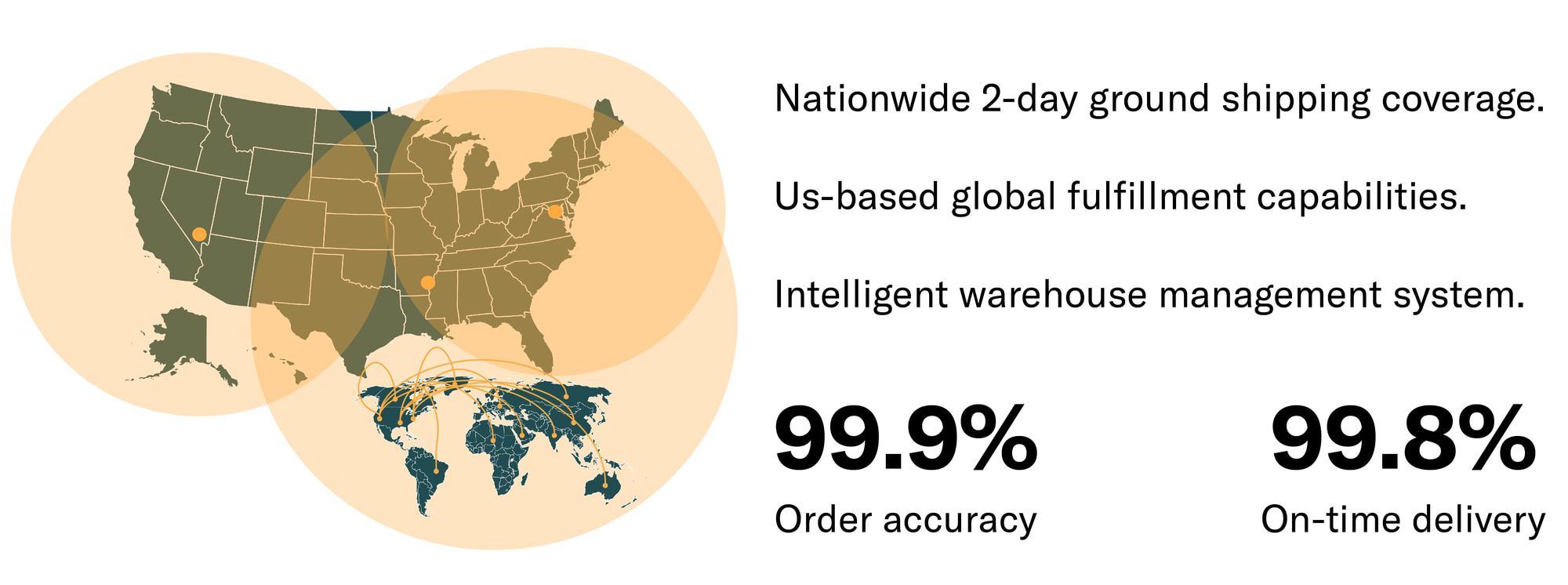
Multi-channel fulfillment Operations: Our high-volume, low-defect fulfillment network was built for modern, multi-channel commerce. Inventory pooling means all sales channels leverage the same warehouse inventory, with real-time availability, nationwide 2-day ground shipping coverage and AI-based forecasting / replenishment.
Market
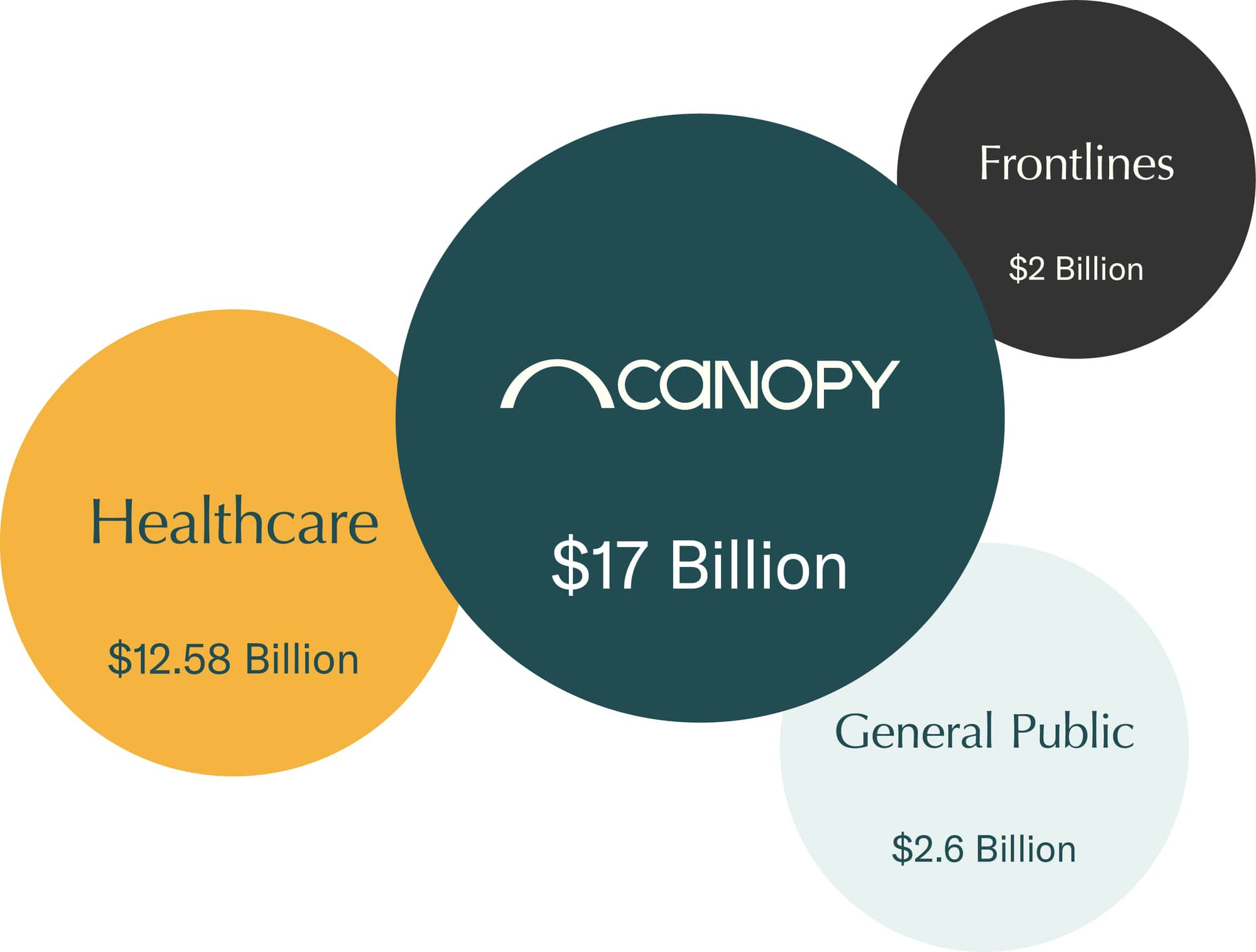
A $17B+ market
With the mass shortage of N95 respirators, we stand to capture a meaningful share of a >$17B market. Beyond the immediate pandemic-related need, institutional buyers are in the market to convert away from disposable respirators to reusable solutions.
While incumbent disposable supply has been steadily increasing to meet demand, a desire to convert away from disposables, matched with few reusable alternatives, creates a major opportunity for Canopy.
Canopy's novel filter design doesn't use globally-scarce meltblown textiles. Every other attempt to design a new mask still calls for the same, unavailable raw material already used in traditional N95s.
Canopy expects to be the first NIOSH-certified reusable respirator designed specifically for medical professionals, and the first new reusable respirator design approved by NIOSH since the pandemic began.
Competition
Desire to abandon disposable N95s met with few reusable alternatives
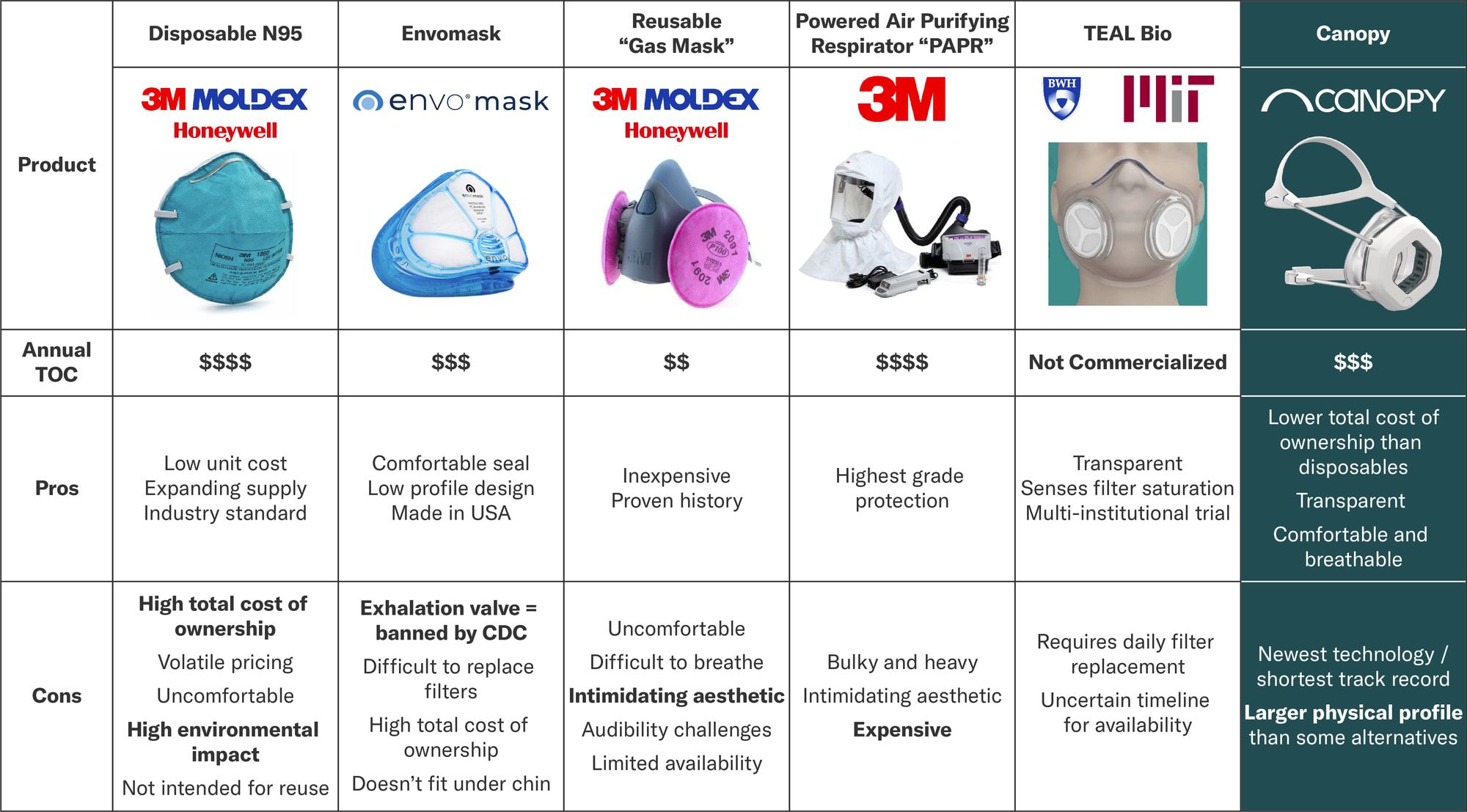
The team at Canopy worked with healthcare professionals to create a respirator that solves for all the pain points of current N95 respirators.
According to recent news reports, Canopy's closest competitor, Envo Mask, introduced their device in Q4 2019 and is selling 1600-2000 units per day. At MSRP, that represents a $46MM - $58MM annual run rate. Canopy's design solves for all of the Envo Mask shortcomings. And ClearMask, a transparent disposable non-N95 surgical mask, is on track to hit $40MM of sales in 2020.
Vision and strategy
Creating sustainable products that increase comfort, safety, and performance
Canopy is the next generation respirator, but it’s also so much more. It’s a pledge we can each make to the well-being of the people and places we love.

Beyond the initial respirator, Canopy’s mission is to protect the world’s well-being by creating sustainable products that increase comfort, safety, and performance. We are exploring additional opportunities to replace single-use products in the clinical setting with superior-performance, more economically efficient alternatives while simultaneously reducing waste.
We’re proud to be a certified B Corporation pending, the highest rating a company our age (less than 12 months) can achieve, and Canopy sales directly address 9 of the 17 UN sustainable development goals. Our forthcoming 501(c)(3) nonprofit, ‘One Canopy,’ will partner with leading NGOs, governments, and public health organizations to distribute Canopy respirators to populations in need, domestic and abroad.

Funding
Capital raise of $1M
To date, we’ve built Canopy exclusively with founder capital from Grove North Ventures, our founder and CEO's investment vehicle.
To supplement our investment, Canopy is raising up to $1.07M to:
- Accelerate sales and marketing initiatives
- Build inventory for rapid fulfillment
- Launch multi-channel paid media campaign
- Pursue product roadmap
- Expand sales and marketing team to support demand
With the current raise, we’ll be able to achieve positive cash flow and set in-motion a substantial inventory build, enabling us to quickly fulfill orders and get Canopy into the hands of those frontline workers who need it most.

Founders
Meet our founder & CEO Joe Rosenberg
Canopy was created by scientists, industrial designers and healthcare professionals who have studied and worked at leading research institutions and innovation centers.

From 2010-2018, Joe was intimately involved in Google’s efforts to revolutionize retail e-commerce, leading the partnership development and management strategy for projects including Google Express and Purchases on Google. He’s partnered with the country’s largest retailers to build innovative business growth strategies.
Joe has created first-of-their-kind partnership deals between Google and organizations in both the retail and financial services industries, including negotiating agreements impacting more than $1B of Google e-commerce transaction volume.
In May 2018 Joe founded Grove North Ventures. Grove provides business incubation and acceleration services, specializing in e-commerce business development. It helps to bring innovative companies and products to the world, building their infrastructure, cultivating strategic partnerships, and developing go-to-market strategies to enable growth early in a company or product lifecycle. Joe frequently advises large companies and top-tier consulting and investment firms in the e-commerce and retail industries as well as on digital transformations. Joe earned a BA with honors from the Gallatin School at NYU in 2010, and is the co-founder of Subject to Change Wine Company, a natural winery in California.
__
A Singapore native, QX was awarded the prestigious Defense Science and Technology Scholarship in 2009 by the Singapore government. After graduating with top honors in 2013 from Johns Hopkins University with a B.S in Biomedical Engineering and MSE in Bioengineering Innovation and Design, QX returned to Singapore to work at DSO National Laboratories, Singapore's national defense research lab.
During his 6-year tenure at DSO, QX specialized in soldier performance, developing products such as the patented A-Lite backpack, which reduced soldier fatigue by properly distributing heavy loads. The backpack received the company's Innovation award, and is currently standard-issue for the Singapore Army.
QX’s team drove DSO’s product development strategy. For his work in shaping the workflow and user interface for the Xentinel counter-drone vehicle, QX was awarded the KINETIC award, the company’s highest internal award.
__
Prior to joining Canopy, Doug spent five years in Operations Innovation management at DaVita, Inc., solving the company’s most pressing process & operations challenges. Doug's work included restructuring and streamlining the customer service model for DaVita's in-house pharmacy, developing and implementing a new electronic health records system to drive enhanced dialysis outcomes, and addressing process and documentation issues that were inhibiting Home Dialysis programs from full billings. Doug's efforts contributed to over $20M annual operational cost reductions, as well as significant improvements to work culture and employee satisfaction.
While at DaVita, Doug also had a leading role in sustainability work on the headquarters campus, and led a team focused on waste reduction to realize sustainable 15% reduction in total waste over two years.
__
Kim currently serves as Senior Vice President, Head of Pension Risk Transfer (PRT) at Athene and is responsible for the management and growth of the company’s PRT business.
Prior to joining Athene, Kim served as Vice President, Head of Distribution & Retirement Solutions for Legal & General’s US PRT business. As a founding member of the business in 2015, Kim led sales, marketing, client solutions and business strategy, growing the business to nearly $4 billion in assets. She previously served as the Deputy Head of Distribution for Legal & General’s US investment business where she worked with corporate pension and insurance clients to implement Liability Driven Investment and fixed income strategies.
Prior to Legal & General, she served as a Director with Cutwater Asset Management, where she helped transform Cutwater to a pension risk management business post-financial crisis where she led the development of pension de-risking strategies and insurance solutions for institutional clients. Kim also led asset-liability management for Cutwater’s $30 billion spread-lending business and held risk roles focused on market risk, hedging, and liquidity management.
A recognized leader in the pension industry, Kim was named to LIMRA’s Rising Stars in Retirement Under 40 List and has published numerous research pieces on pension risk management.
Kim holds a BBA in public accounting and an honors degree from the Pforzheimer Honors College at Pace University where she graduated magna cum laude. She also holds an MBA from Marist College.


 Oops! We couldn’t find any results...
Oops! We couldn’t find any results...






















