Neopenda, a Chicago-based public benefit corporation, is now raising funds in an equity crowdfunding campaign. The campai...
 Oops! We couldn’t find any results...
Oops! We couldn’t find any results...
 Oops! We couldn’t find any results...
Oops! We couldn’t find any results...

Neopenda

Open for investment
Documents
 Form C
SEC.gov
Form C
SEC.gov
Deal highlights
- Medical device company with potential to save up to 2.6M newborn lives a year in emerging economies
- $7.1B target market by 2023: Medical devices in Africa
- Patent pending and additional provisional patents filed
- Founders are biomedical engineers out of Columbia University
- Techstars Chicago Class of 2018
- $770k raised to date from Columbia University, Cisco, Vodafone, ADAP Capital, Techstars, and others
- A for-profit Public Benefit Corporation
- Become part of Neopenda’s journey for as little as $100, and see how a little goes a long way in saving lives
We Are Neopenda.
Our name comes from “neo” - for neonates, and “penda” - Swahili for love.
Preventable Newborn Mortality: 3 million newborns are lost in developing countries each year
In fact, a newborn born in a developing country like Uganda has a nine times poorer chance of surviving its first month than one born in the United States.

It doesn’t have to be this way. WHO data shows that 80% of newborn deaths are preventable. The primary causes are complications of preterm birth, birth asphyxia, and infections. Although these conditions are treatable, many newborns do not receive the attention they need because facilities are overcrowded, understaffed, and ill-equipped. Nurses in these facilities don’t have the tools they need to provide high quality neonatal care. As a result, babies are suffering—avoidably.
Neopenda’s Solution: A 4-in-1 Vital Signs Monitor
At Neopenda, we believe that all communities deserve access to innovative, life-saving health technology. There exists a massive opportunity to create appropriately designed medical devices for a largely untapped market in emerging economies, and to do so in a way that can improve quality of care for millions of patients in need.

Our first product is a wearable 4-in-1 vital signs monitor designed to enable more responsive and appropriate medical care of newborn infants in resource constrained hospital facilities. The patent-pending device uses reflectance pulse oximetry and temperature sensors, and is worn in a reusable band. It continuously measures:

The vitals from many devices are wirelessly displayed on a tablet, where the nurse can see what’s going on with all the babies. She gets alerted when vitals go outside the healthy range, so that she knows where her attention is needed and give patients the best chance to survive and thrive.
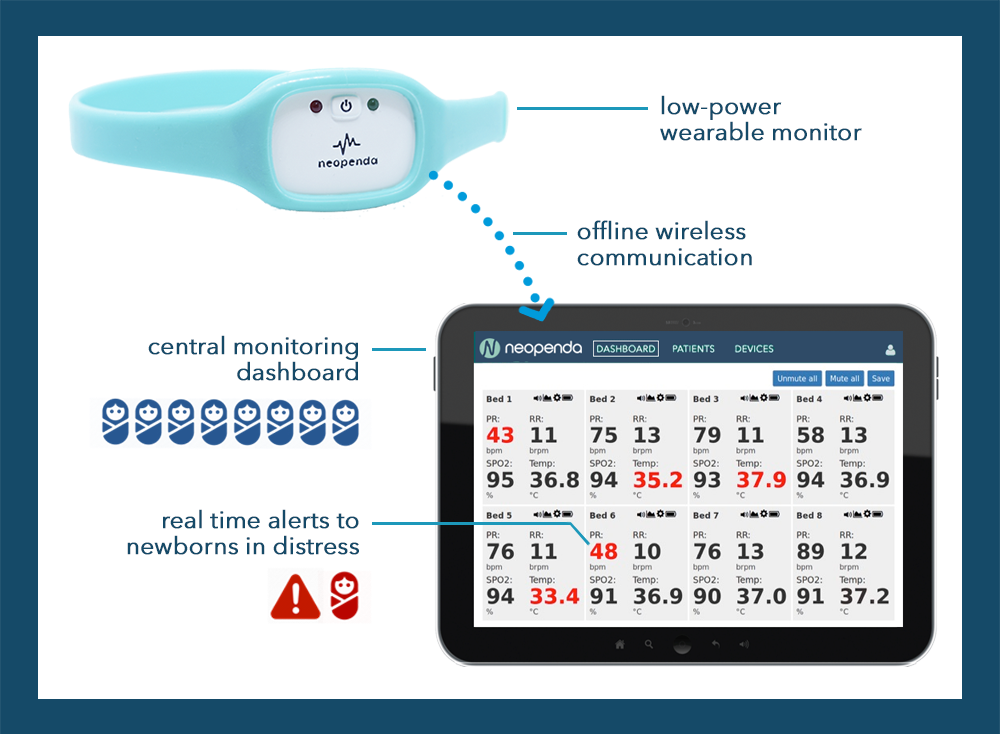
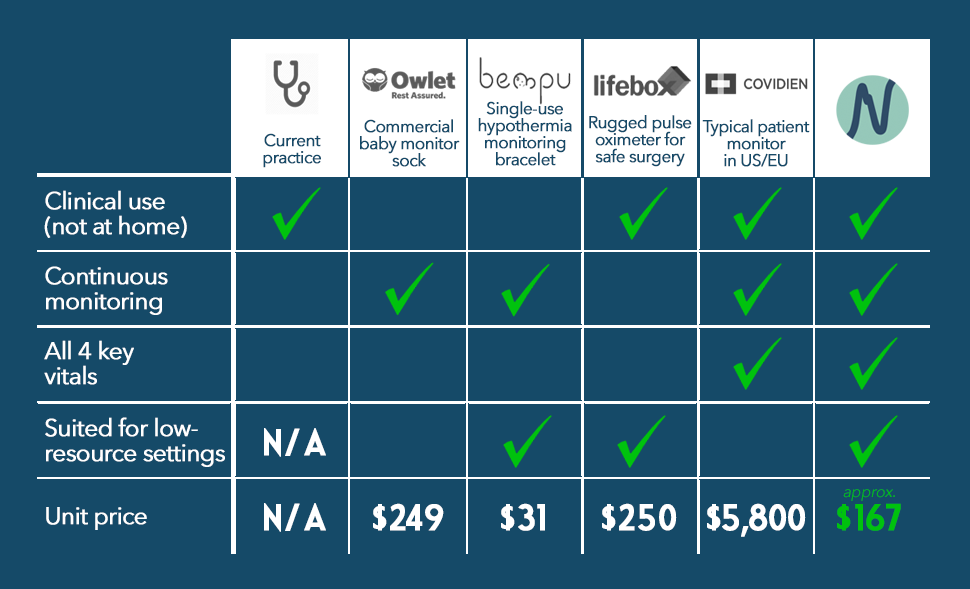
Today, nurses in these facilities have to check babies’ vitals by hand. However, hospitals don’t have enough staff to safely monitor newborns as often as they need to be. Neopenda’s product is the only option known to us that continuously measures all crucial vital signs, and unlike other patient monitors or baby monitors, is tailored for use in low-resource settings. We’ve designed our product to be affordable, easy to use, and withstand tough environments for a long time. It doesn’t require continuous stable power or wireless internet.
Hear From Our Stakeholders

Traction and Partners
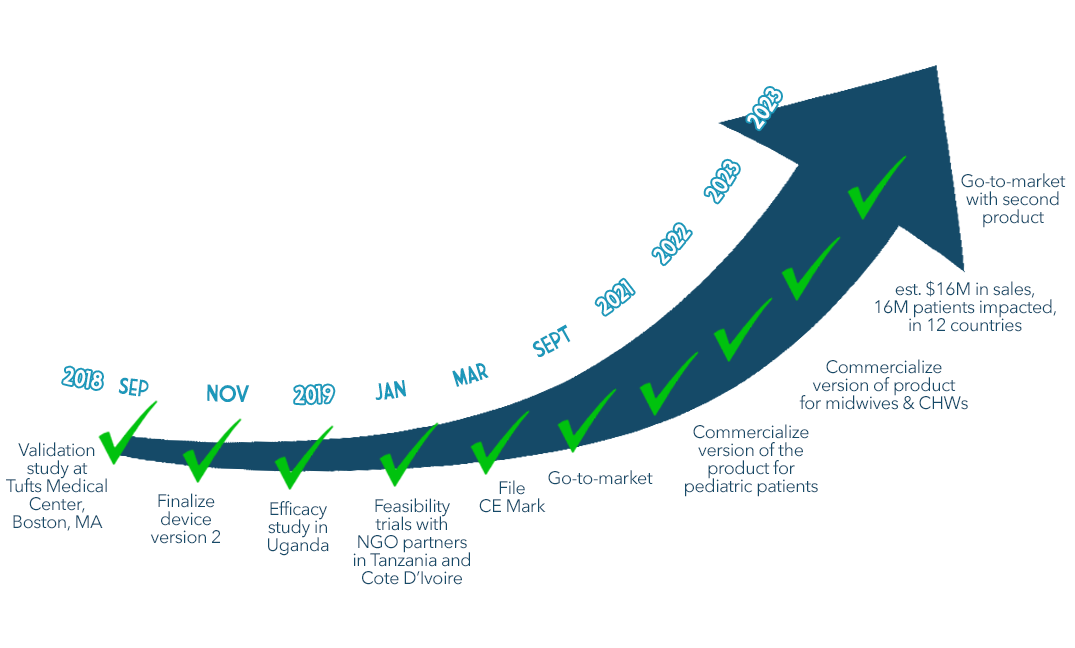
We’re dedicated to getting this right. We’ve spent the last three years in hospitals in Uganda learning from stakeholders and testing the product with nurses and doctors through our human-centered design process.

We’ve already joined forces with a diverse set of partners across the value chain:

Recognition

Our Business Model

We will sell our product in packages containing 15 wearables, 1 tablet, and all associated software, power supplies, and accessories. This package provides comprehensive monitoring for an entire 15-bed newborn unit for less than half the price of a single machine traditionally used in the U.S.
Gross profit margin on the package: 60-70%
System installation, training, and product support is included
Dual distribution modes for rapid market entry:
Selling direct to hospitals via in-country wholesale distributors such as the Joint Medical Stores in Uganda
Partnering with aid agencies such as Doctors Without Borders, whose international presence will help us rapidly scale
Additionally, the detailed frontline health data we’re collecting is of great interest to parties like NGOs and Ministries of Health. We’re currently working on a strategy to aggregate, analyze, and monetize this valuable data.
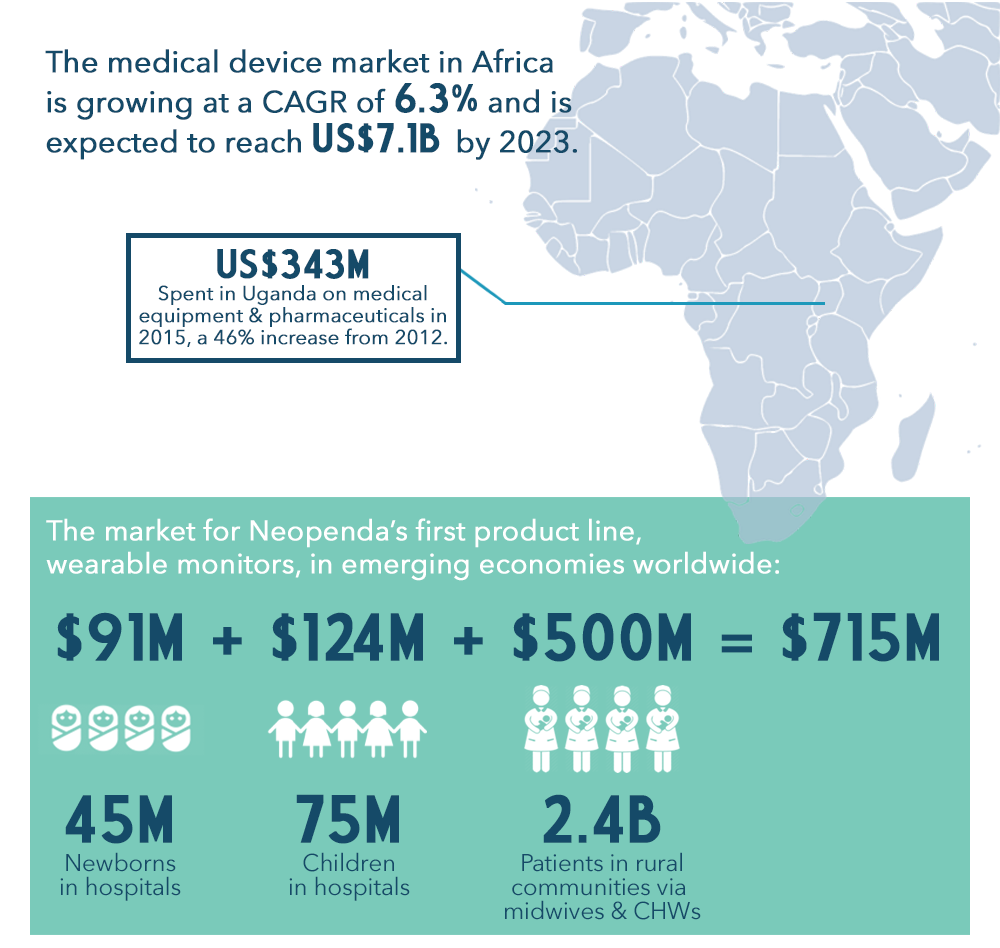
The Market: An Untapped Opportunity in Africa
Competition
Funding History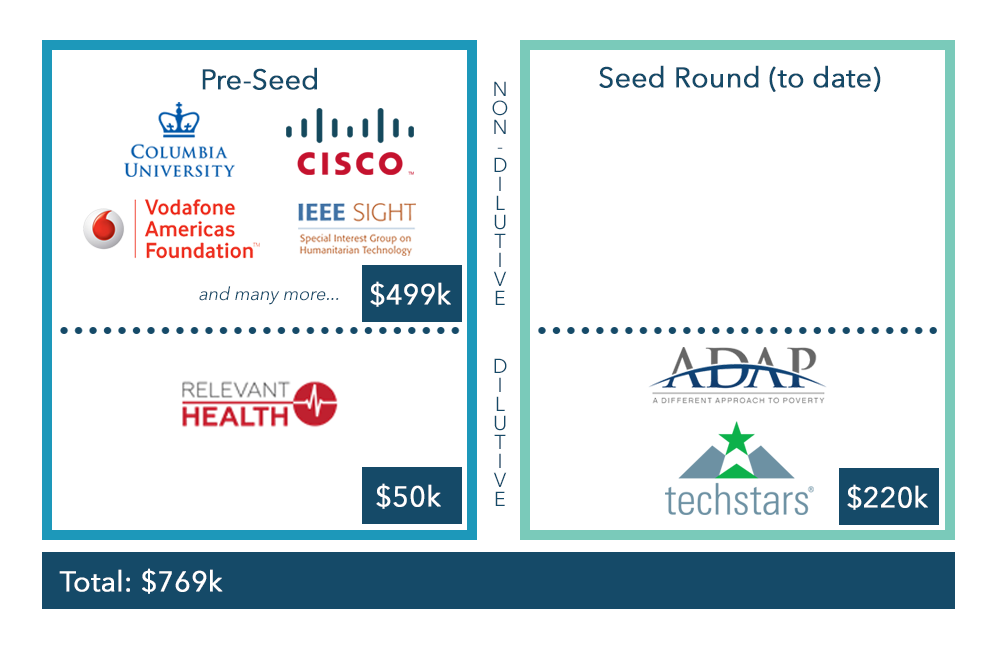

Team Neopenda
We are a team of biomedical engineers and public health experts who care deeply about saving lives and transforming the way health tech is approached in emerging markets.
Co-founders Sona (CEO) and Teresa (CTO) began Neopenda in 2015 as graduate students in Biomedical Engineering at Columbia University, after witnessing first hand in Uganda the massive opportunity to sustainably improve health outcomes for vulnerable patients. We also have Dorothy and Michael based in Kampala, Uganda, working on program activities and research activities, respectively.

What's Next
We have a lot of exciting milestones to accomplish in the next year, and we need your help to get there! Here’s what we will do next:
We will be using your investment dollars to bring our solution to market and accelerate our growth:
- $25,000 will fund a clinical study in Uganda
- $100,000 will fund 3 pilot studies in Uganda and 1 in Tanzania
- $200,000 will fund expansion to 6 countries in Sub-Saharan Africa plus India
Join Us!
Neopenda is pioneering health tech in emerging markets. We're starting with a vital signs monitoring tool to help save vulnerable newborn lives.
We invite you to join us, as we create medical solutions for where they’re needed most!
Deal terms
$6,000,000
The maximum valuation at which your investment converts
into equity shares or cash.
Learn more
20%
If a trigger event for Neopenda occurs, the discount provision
gives investors equity shares (or equal value in cash) at a reduced price.
Learn more.
$1.07M
Neopenda must achieve its minimum goal of $25K before the deadline. The maximum amount the offering can raise is $1.07M.
Learn more
Learn more
Crowd SAFE
A SAFE allows an investor to make a cash investment in a company, with rights to receive certain company stock at a later date, in connection with a specific event.
·
Learn more
Documents
 Form C
SEC.gov
Form C
SEC.gov
Bonus perks
- We will give you a personal shout out on our website (with your consent)!
- All of the above + we’ll add your name to our “plaque” of supporters in our office
- All of the above + you’ll receive a Neopenda t-shirt.
- All of the above + a Neopenda photo book telling the story of our work in Uganda.
- All of the above + dinner with the founders at our HQ in Chicago.
About Neopenda
Neopenda Team
Everyone helping build Neopenda, not limited to employees
Press
This site (the "Site") is owned and maintained by OpenDeal Inc., which is not a registered broker-dealer. OpenDeal Inc. does not give investment advice, endorsement, analysis or recommendations with respect to any securities. All securities listed here are being offered by, and all information included on this Site is the responsibility of, the applicable issuer of such securities. The intermediary facilitating the offering will be identified in such offering’s documentation.
All funding-portal activities are conducted by OpenDeal Portal LLC doing business as Republic, a funding portal which is registered with the US Securities and Exchange Commission (SEC) as a funding portal (Portal) and is a member of the Financial Industry Regulatory Authority (FINRA). OpenDeal Portal LLC is located at 149 E 23rd St #1314, New York, NY 10010, please check out background on FINRA’s Funding Portal page.
All broker-dealer related securities activity is conducted by OpenDeal Broker LLC, an affiliate of OpenDeal Inc. and OpenDeal Portal LLC, and a registered broker-dealer, and member of FINRA | SiPC, located at 1345 Avenue of the Americas, 15th Floor, New York, NY 10105, please check our background on FINRA’s BrokerCheck.
Certain pages discussing the mechanics and providing educational materials regarding regulation crowdfunding offerings may refer to OpenDeal Broker LLC and OpenDeal Portal LLC collectively as “Republic”, solely for explanatory purposes.
Neither OpenDeal Inc., OpenDeal Portal LLC nor OpenDeal Broker LLC make investment recommendations and no communication, through this Site or in any other medium should be construed as a recommendation for any security offered on or off this investment platform. Investment opportunities posted on this Site are private placements of securities that are not publicly traded, involve a high degree of risk, may lose value, are subject to holding period requirements and are intended for investors who do not need a liquid investment. Past performance is not indicative of future results. Investors must be able to afford the loss of their entire investment. Only qualified investors, which may be restricted to only Accredited Investors or non-U.S. persons, may invest in offerings hosted by OpenDeal Broker.
Neither OpenDeal Inc., OpenDeal Portal LLC nor OpenDeal Broker LLC, nor any of their officers, directors, agents and employees makes any warranty, express or implied, of any kind whatsoever related to the adequacy, accuracy or completeness of any information on this Site or the use of information on this site. Offers to sell securities can only be made through official offering documents that contain important information about the investment and the issuers, including risks. Investors should carefully read the offering documents. Investors should conduct their own due diligence and are encouraged to consult with their tax, legal and financial advisors.
By accessing the Site and any pages thereof, you agree to be bound by the Terms of Use and Privacy Policy. Please also see OpenDeal Broker’s Business Continuity Plan and Additional Risk Disclosures. All issuers offering securities under regulation crowdfunding as hosted by OpenDeal Portal LLC are listed on the All Companies Page. The inclusion or exclusion of an issuer on the Platform Page and/or Republic’s Homepage, which includes offerings conducted under regulation crowdfunding as well as other exemptions from registration, is not based upon any endorsement or recommendation by OpenDeal Inc, OpenDeal Portal LLC, or OpenDeal Broker LLC, nor any of their affiliates, officers, directors, agents, and employees. Rather, issuers of securities may, in their sole discretion, opt-out of being listed on the Platform Page and Homepage.
Investors should verify any issuer information they consider important before making an investment.
Investments in private companies are particularly risky and may result in total loss of invested capital. Past performance of a security or a company does not guarantee future results or returns. Only investors who understand the risks of early stage investment and who meet the Republic's investment criteria may invest.
Neither OpenDeal Inc., OpenDeal Portal LLC nor OpenDeal Broker LLC verify information provided by companies on this Site and makes no assurance as to the completeness or accuracy of any such information. Additional information about companies fundraising on the Site can be found by searching the EDGAR database, or the offering documentation located on the Site when the offering does not require an EDGAR filing.
To help the government fight the funding of terrorism and money laundering activities, Federal law requires all financial institutions to obtain, verify, and record information that identifies each person who opens an account. Therefore, when you use the Services we will ask for your name, address, date of birth, and other information that will allow us to identify you. We may also ask to see your driver's license, passport or other identifying documents.
Republic and its affiliates are not and do not operate or act as a bank. Certain banking services are provided by BankProv, member FDIC / member DIF. Digital (crypto) assets and investment products are not insured by the FDIC, may lose value, and are not deposits or other obligations of BankProv and are not guaranteed by BankProv. Terms and conditions apply.

Made in SF/NYC





















