Por Angélica Pineda Liza Velarde hizo historia el pasado 7 de mayo. La cofundadora de Delee, start-up de salud acelerada ...
Problem

According to the WHO, in 2018, the global cancer burden increased to 18.1 million new cases, causing 9.6 million deaths. Despite the many advances in the matter of treatments, it is estimated that in 2040, cancer cases will increase to 29.5 million, and approximately 16.3 million will die because of it.
One of the main reasons cancer has such a high mortality rate is due to the current lack of clinical tests with sufficient sensitivity and specificity to enable a timely diagnosis of this disease. In addition, the scarcity of technological resources to provide effective monitoring of the treatments applied, significantly reduces the patients’ chances of survival. The isolation of circulating tumor cells from the blood is a novel alternative to address these issues.
Finding the needle in the haystack
For most types of cancer, when a person develops a tumor, even if it hasn’t spread yet, it releases malignant cells into the bloodstream, known as circulating tumor cells or CTCs, which play a key role in establishing metastasis in other organs.
In the past few years, it has been demonstrated that CTCs can be isolated from blood, facilitating their enumeration and analysis. This could enable early cancer diagnosis and allow the proper monitoring of the therapies’ effectiveness when being administered to the patient. However, the isolation of these malignant cells from blood represents a major technological challenge due to their rareness. You can find ~37.5 billion cells in 7 ml of blood but, in a cancer patient, only 1 to 50 of those would be tumor cells; this is equivalent to identifying just a couple of people within the population of 5 Earths.

Even though there are cell sorting methods such as flow cytometry, density gradient centrifugation, among others, they do not have sufficient sensitivity and specificity to isolate CTCs. To be able to detect these rare cells, current methods would have to be ~100X more sensitive.
Solution
CytoCatch: detecting circulating tumor cells
At Delee we have created the CytoCatch™, a device that with a simple blood extraction starts a rapid process to successfully isolate circulating tumor cells.
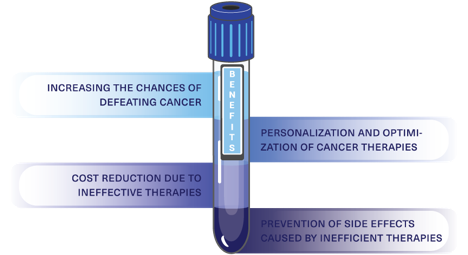
Unlike other tests, CytoCatch™ possesses the required sensitivity and specificity to analyze the CTCs genetic features, as well as predictive and therapeutic markers expressed on them. Facilitating the early detection of cancer and enabling the personalization and optimization of each patient’s treatment. Because of this, patients and their families will be able to save time, reduce costs, prevent side effects of inefficient therapies, and more importantly, increasing their odds of defeating cancer.


Product
Able to diagnose and monitor treatment
Once blood samples have been previously extracted by conventional methods, CytoCatch™ isolates the CTCs and automatically performs the necessary procedures to stain the captured cells with fluorescent antibodies, in order to discriminate the tumor ones from the other cellular components of the blood. Later, the integrated imaging system, which possesses artificial intelligence algorithms, analyzes the malignant cells, based on their morphology and the expression of specific antigens.
Our device is capable to individually collect tumor cells by implementing micromanipulation techniques, enabling to perform molecular analysis and assess the genetic characteristics of the CTCs. Finally, a report with the results will be given to the physician.

Clinical applications
The CytoCatch™ has the specificity and sensibility required to successfully isolate and analyze circulating tumor cells from blood, deriving on the following clinical applications:

Benefits
The early detection of cancer and the personalization of treatments for each patient can be translated into invaluable benefits for them and their families; the most important, and the main reason for our work is to increase their chances of defeating cancer and saving their lives. Monitoring the effectiveness of cancer therapies could also reduce the overall investment cost and the negative side effects caused by drugs that would not be effective.
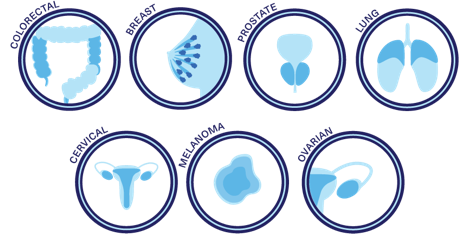
For what types of cancer?
There is clinical evidence that supports CTCs as a powerful biomarker that can be used to early detect cancer and to monitor the effectiveness of the therapies that are being administered to patients with cancer, such as prostate, breast, colorectal, lung, cervical, melanoma, ovarian, among others. From the types mentioned above, the first four, alone, are responsible for more than a third of the defunctions occasioned by this disease.
Traction
Pre-sales with a value of $1.4M
We’ve gotten great traction since our pre-commercial launch of the CytoCatch™ device. To date, presales with a value of $1.4 million USD in potential revenue have been secured from research centers of various hospitals, to use our technology prior to FDA clearance. In addition, we’ve received attention from major research institutions and industry partners, including Harvard University, Stanford University, TEC de Monterrey, UANL, among others.
Delee has partnered with Sheppard Mullin, one of the top multinational intellectual property firms, to develop a strong IP strategy that strengthens our business globally. We’ve submitted both an international patent application PCT and provisional patent applications that encompasses hardware, software, biological protocols, applications, and data analysis methods.

Our work has been published in top peer-reviewed journals, and has been presented in various international conferences. Recently, our research was awarded by the Fundació Puigvert, a highly renowned international institution in the urology field, as the best clinical work presented at the congress of the American Confederation of Urology, this validates the interest of the medical community in our technology.
Customers
A tool for researchers and pharma
Prior to FDA clearance, Delee plans to commercialize the CytoCatch™ as a research tool with pharmaceutical companies and researchers as our main customers. Once the technology obtains the FDA approval, it can be commercialized, as an in vitro diagnostic medical device, to hospitals and laboratories.
Business model
Recurring revenue through consumable and reagent sales
Prior to FDA clearance, the razor and blades business model will be followed, obtaining recurrent revenue by selling the necessary reagents and consumables to perform each test. This model will be maintained after obtaining FDA approval for the commercialization of our technology.

Market
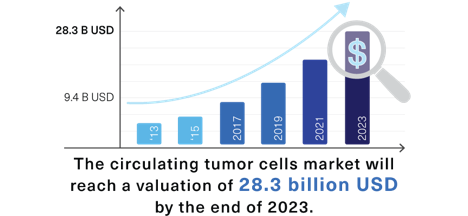
A $28.3B market by 2023
According to the report published by Market Research Future, in 2017, the global market for circulating tumor cells was valued at 9.39 billion USD, and it is estimated that by the end of 2023 it will reach a valuation of 28.3 billion USD. The increase in market valuation will be driven by the increment of the number of new cases and the expected mortality over time.
At Delee we are aiming to initially validate the CytoCatch™ in prostate and breast cancer and to further expand the validation to other types, such as lung and colorectal cancer. Because these four types of cancer are responsible for 40% of the new cases worldwide, triggering the deaths of 3.6 million people a year.
Competition
A more effective way to capture CTCs
Most tests employed as auxiliaries for cancer diagnosis and therapies’ monitoring measure tumor marker levels in blood, typically proteins like PSA, CA 125 and AFP for prostate, ovarian and liver cancer, respectively. Nevertheless, those tests lack sufficient sensitivity and specificity, leading to an elevated number of false positive and negative results.
The isolation and analysis of CTCs is a relatively new practice, and physicians are starting to recognize all its potential benefits. Most CTCs technologies, including the CellSearch®System, which is considered the gold standard, rely on the existence of specific proteins on the cell membrane to capture CTCs. However, cancer cells are incredibly heterogeneous; when entering the bloodstream, they undergo a process in which the proteins used to capture CTCs are downregulated, limiting the capture of tumor cell subpopulations with diminished expression of there specific proteins, thereby losing valuable information. Our technology changes the norm by isolating CTCs irrespective of the level of proteins expressed in their membranes, allowing us to capture cells that other technologies can’t.
Vision and strategy
Launching the CytoCatch in 2020

Funding
Delee is a Y Combinator company
Delee has secured over $1.3 million in funding from Wall Street, Silicon Valley, and healthcare investors.
Founders

Co-Founder and acting CEO at Delee Corp., Liz Velarde is a Y Combinator alumna and graduated summa cum laude from the Tecnologico de Monterrey with a Bachelor of International Business degree. Throughout her career she has raised over $1.3M through investments, government funds, and multiple awards. At Delee, Velarde has been responsible for the development and execution of the company's strategic plans. In addition, she is responsible for leading a multidisciplinary team that has created a technology successfully tested on patients with prostate cancer, for the isolation and analysis of Circulating Tumor Cells, with pre-sales that exceed $1.4M. She has also enabled the establishment of strong relations with top hospitals and research centers. Velarde's outstanding work has been highly regarded by international institutions such as Cartier Women's Initiative Awards and WeXchange (from the Inter-American Development Bank). In October 2019, she was acknowledged as one of the 50 most relevant people who are transforming Mexico, and was invited as a speaker on various international panels about cancer and extrepreneurship such as WeXchange 2019 and The Economist: War on Cancer LATAM 2019. She also lead and supervised the development of the Zen FluidicsTM scientific line, a group of products that enables researchers to develop their own microfluidic applications.
Juan Felipe Yee is a co-Founder, and acting Chief Operating Officer (COO) at Delee Corp., where he also actively participates in the development and execution of the company’s strategic plans. Yee is a Y Combinator alumnus, and completed his B.Sc. in Biomedical Engineering and his M.Sc. in Electronic Engineering at the Tecnológico de Monterrey. He has spent over a decade working and collaborating in the development of various medical devices and biosensors such as a high intensity phototherapy LED source to treat hyperbilirubinemia in newborns, substrates made from carbon nanofiber mats coated with gold nanoparticles for the detection of specific molecules in simple solutions by SERS spectroscopy, and microfluidic devices for cell isolation based on antigen-antibody interactions, inertial forces, and dielectrophoresis. Yee has collaborated with the Biomedical Engineering Group at Tecnológico de Monterrey in projects related to biomaterial and tissue engineering and the development of organ-on-chip systems. He is a co-creator of the CytoCatch™, a device that isolates and analyzes circulating tumor cells from blood samples, and the Zen Fluidics™ scientific line, which targets the microfluidics market.
Alejandro Abarca is the co-Founder and acting Chief Technology Officer at Delee Corp., where he is responsible for the design and execution of Delee’s strategic plans regarding R&D and product development. Abarca is a Physicist, a Y Combinator, Singularity University, and a Royal Academy of Engineering LIF alumnus. He has over ten years of experience developing and producing medical devices and biosensors such as a microfluidic device for the isolation of rare cell subpopulations based on dielectrophoretic separation, manufacturing methods for embedding metal electrodes onto thermoplastics for microfluidic applications, and an automated imaging system based on fluorescence to study cellular properties. Abarca also has collaborated in projects related to bioprinting and point-of-care applications with various research groups at Tecnológico de Monterrey. His areas of expertise include microfabrication, manufacturing techniques for mass production, optics, and cell separation based on physical properties. He is a co-creator of the Zen Fluidics™ scientific line and the CytoCatch™ system, a device that isolates and analyzes circulating tumor cells from blood samples for the early diagnosis and monitoring of the efficiency of cancer therapies.
Directors

Joost Leeflang has over 20 years experience in successfully driving top and bottom line growth for consumer and professional business in and across multiple geographies. He served as Senior VP and Global Head of Commerce responsible for Global Sales and Marketing for the Medical Imaging Business of Philips Healthcare. Prior to that, Joost had occupied several key positions in the Philips Organization, including Senior VP and CEO of Philips Electronics Central and Eastern Europe, Senior VP and General Manager of Philips Lightning in Europe, Senior VP, CEO and General Manager of Philips Electronics RUC in the Healthcare & Consumer Lifestyle area, amongst others. He was the COCIR President, the European Trade Association representing the medical imaging, health ICT, and electromedical industries. Joost completed an Executive Program in Strategy and Organization at the Stanford Business School and a Master in Business Economics at the University of Groningen.
Scientific and Medical Advisory Board

Mario Moisés Álvarez is the Director of the Biomedical Engineering Group at Tecnológico de Monterrey. Alvarez has been a Visiting Professor at the Harvard-MIT Health Sciences and Technology at BWH, Harvard Medical School, and a Visiting Professor at the Microsystems Technology Laboratories (MTL) at the Massachusetts Institute of Technology. His research interest is focused on the integration of bio-, micro-, and nanotechnologies into tissue, biomedical, and biopharmaceutical engineering applications. His current academic production is mainly focused on bioprinting, biomaterial and tissue engineering, development of organ-on-chip systems, point of care diagnostics, and biopharmaceutical engineering. Professor Alvarez completed an industrial postdoctoral stay at the Institute of Pharmaceutical Technology at Bristol-Myers Squibb Company, USA. He holds a Ph.D. in Chemical and Biochemical Engineering from Rutgers University, a M.Sc. in Chemical and Biochemical Engineering from Rutgers University, a M.Sc. in Chemical Engineering from Tecnológico de Monterrey, and a B.Sc. in Biochemical Engineering from Tecnológico de Monterrey. Alvarez has published 85+ papers in international journals. He has been granted with three US patents and twelve Mexican patents. Alvarez is a permanent member of the Mexican Academy of Sciences and a member of the National Research System, where he has been awarded with the highest ranking for Mexican researchers (SNI Level III).

David Mohler is an Orthopedic Surgical Oncologist, Clinical Professor, and Chief of the Musculoskeletal Tumor Service Department of Orthopedic Surgery and Sports Medicine from Stanford University Medical Center. Mohler received his B.Sc. from Stanford University and his M.D. from Cornell University Medical College. He completed an internship in General Surgery at UCLA, a residency in orthopedic surgery at the Hospital for Special Surgery, and a Metabolic Bone Disease/Bone Tumor fellowship at the Memorial Sloan-Kettering Cancer Center. With over three decades of experience in the field, he has authored and co-authored more than 35 papers in peer-reviewed journals, 5 book chapters, and collaborated in various research projects. Mohler is currently a member of several prestigious associations such as the Orthopedic Research Society, the American Academy of Orthopedic Surgeons, the Musculoskeletal Tumor Society, among others. He has an American Board of Orthopedic Surgery certification and has been honored with awards like the Alpha Omega Alpha, The National Research Service Award and the Saul Halpern, MD Orthopedic Educator Award.

Grissel Trujillo de Santiago is a Professor at the School of Engineering and Sciences at Tecnológico de Monterrey. She was trained as a Chemistry & Pharmacy Biologist at Universidad Autónoma de Nuevo León (summa cum laude); she obtained her M.Sc. degree in Biotechnology (summa cum laude) from Tecnológico de Monterrey; and she received her Ph.D. degree in Biotechnology from Tecnológico de Monterrey. Dr. Trujillo de Santiago conducted research in Material Sciences at the Università degli Studi di Napoli Federico II (Naples, Italy) during her Ph.D. program. She was a Postdoctoral Research Fellow at Dr. Khademhosseini’s Lab in the Harvard-MIT Health Sciences and Technology Division for 2 years and the Microsystems Technology Laboratories at MIT, where she collaborated in several projects related to biomaterials engineering, bioprinting, tissue engineering, organ-on-chip systems, and point-of-care applications. She has published 30 papers in Indexed International Journals and one book chapter, has been awarded with a Mexican patent, and has more than thirty participations in International Conferences. She was a recipient of the For Women in Science Award from the L’Oreal-UNESCO-CONACyT-AMC foundation in 2019. Her main area of interest and expertise is the design and fabrication of biomaterials suitable for biomedical applications such as Tissue Engineering and organs-on-chip. Dr. Trujillo de Santiago research is currently focused on the development of novel platform-technologies based in chaos to produce 3D micro- and nanostructures in fluid biopolymers.

Lauro S. Gómez is a Urologist and Research Director of the Department of Urology at the University Hospital “Dr. José Eleuterio González”. He has over 30 years of experience in the oncological, pediatric and male infertility fields, has authored and co-authored over 60 papers in peer-reviewed journals, and is a reviewer of various specialized medical journals. He is President of the School of Urologists of Nuevo León, a permanent member of the Mexican Academy of Surgery and the Mexican Society of Urology, and a member of the National Research System. Lauro completed his M.D. and specialty in urology at the Universidad Autónoma de Nuevo León, and received his subspecialty in pediatric urology, andrology and microsurgery from the Puigvert e Institut Dexeus Foundation of the Autonomous University of Barcelona and Necker Hospital of the Paris University.



 Oops! We couldn’t find any results...
Oops! We couldn’t find any results...






















